Germline mutations in the shelterin component protection of telomeres 1 (POT1) were recently found to be associated with familial chronic lymphocytic leukemia (CLL), melanoma, glioma, and several other familial cancer syndromes. The role of POT1 mutations in myeloid neoplasms and other hematologic malignancies, however, remains unknown. To explore the role of POT1 variants in hematologic neoplasms, we analyzed POT1 variants in 3323 consecutive patients who underwent next-generation sequencing (NGS) of a panel of hematologic malignancy-associated genes at our institution and characterized the clinical and pathological characteristics of patients with germline and somatic POT1 mutations.
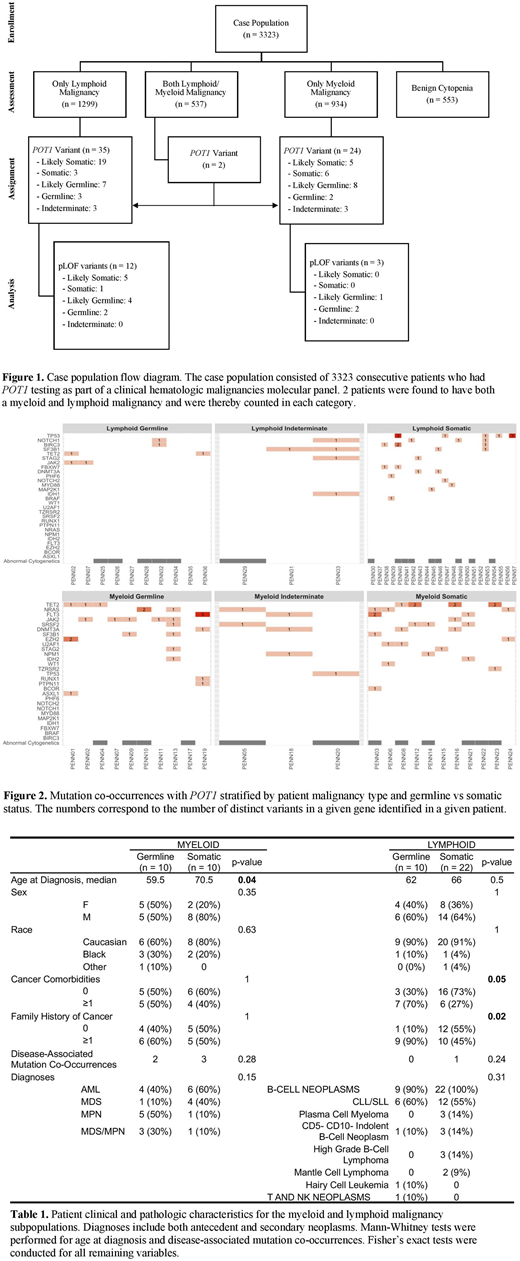
Of 3323 consecutive patients who underwent NGS, 2770 patients had a hematologic malignancy (lymphoid n = 1299, myeloid n = 934, and both lymphoid and myeloid n = 537), while 553 patients were evaluated for non-malignant cytopenias. All 57 patients (2.06%) carrying either a POT1 disease-associated variant or variant of uncertain significance had a hematologic malignancy compared to no identified POT1 variants in 553 patients with benign cytopenias (OR = 23.5, p < 0.001), suggesting that the presence of POT1 variants was predictive of a hematologic malignancy. Of 57 patients, 33 had lymphoid malignancies, 23 had myeloid neoplasms, and 2 had a lymphoid and myeloid neoplasm (Fig 1).
Patient variants were classified as germline or somatic using constitutional DNA sequencing, POT1 emergence/disappearance over time, or POT1 maintenance in remission. In the absence of these data, likely germline or likely somatic designations were made by assessing variant allele frequencies against clinical/pathologic characteristics. 18 patients (33%) were found to have germline or likely germline POT1 variants (29% and 42% in the lymphoid and myeloid malignancy groups, respectively). Another 6 patients (11%) had variants whose germline status could not be determined. Of the 17 unique germline POT1 variants, 10 were missense and located within mapped functional protein domains, while 7 were classified as predicted loss-of-function (pLOF) due to a disruption of start, premature stop, frameshift, or spice site alteration. Patients with hematological malignancies had a ~5-8x increased odds of having a germline pLOF POT1 variant compared to cancer-free individuals in the Genome Aggregation Database (gnomAD, n = 113,108 exomes, OR = 7.5, p < 0.001) or in the Penn Medicine BioBank (PMBB, n = 7877, OR = 5.0, p = 0.010), with a prevalence of 0.25% compared to 0.03% and 0.05%, respectively. Germline pLOF POT1 variants were significantly more enriched in patients with myeloid (gnomAD: OR = 6.1, p = 0.02) and lymphoid (gnomAD: OR = 9.8, p < 0.001; PMBB: OR = 6.5, p = 0.004) malignancies.
In 33 patients with lymphoid malignancies and POT1 variants, the most common diagnoses were CLL/SLL (n = 21, germline n = 6, somatic n = 12), CD5- CD10- indolent B cell neoplasms (n = 4, germline n = 1, somatic n = 3), and multiple myeloma (n = 3, all somatic) (Table 1). Lymphoid malignancies with a germline POT1 variant had a relative paucity of additional mutations; in contrast, somatic POT1 variants frequently co-occurred with other mutations, most commonly with TP53 (Fig 2, n = 5, 23%). Among 23 patients with myeloid malignancies, patients with germline POT1 variants developed malignancies at a significantly younger age compared to those whose POT1 variants were somatic (median age 59.5 vs 70.5 years, p = 0.04). The most common diagnosis in patients with myeloid neoplasms carrying germline POT1 variants was MPN (germline n = 5, somatic n = 1). AML, MDS/MPN, and MDS occurred in 4, 3, and 1 patients respectively. All patients with myeloid neoplasms had additional disease-associated mutations, with the most common co-occurring variants in TET2 (n = 7), JAK2 (n = 6, co-occurring with 50% of germline POT1 myeloid variants), and NRAS (n = 6).
In conclusion, this is the first comprehensive analysis of POT1 variants in an unselected hospital-based population undergoing molecular testing for variants associated with hematologic malignancies. Our results show that the presence of POT1 variants is predictive of having a hematologic neoplasm and that over 30% of POT1 variants in hematologic malignancy patients are germline. Our study expands the spectrum of POT1-associated familial neoplasms and highlights the needs for better recognition of familial hematologic cancer syndromes.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal